International Day For The Preservation Of The Ozone Layer
- 16 Sep 2019
- On 16th September, 2019, World Ozone Day was celebrated under the theme- 32 Years and Healing, to mark the day back in 1987 when the Montreal Protocol was signed.
- World Ozone Day has been celebrated since 1994 and was established by the United Nations General Assembly. The day is mainly intended to spread awareness of the depletion of the Ozone Layer and search for solutions to preserve it.
Ozone Timeline
Vienna Convention
- The Vienna Convention for the Protection of the Ozone Layer which is considered precursor to the Montreal Protocol was adopted in 1985.
- It aimed to protect human health and the environment against the adverse effects resulting from modifications of the ozone layer.
- It was followed shortly afterwards by the adoption of its Montreal Protocol on Substances that Deplete the Ozone Layer in 1987.
Montreal Protocol
- Signed in 1987, it was the first step in international efforts to protect stratospheric ozone.
- Under this, all parties have specific responsibilities related to the phase out of the different groups ozone depleting substances(ODS), control of ODS trade, annual reporting of data, national licensing systems to control ODS imports and exports, and other matters. Developing and developed countries have equal but differentiated responsibilities, but most importantly, both groups of countries have binding, time-targeted and measurable commitments.
- The substances controlled by the treaty are listed in Annexes A (chlorofluorocarbons(CFCs)halons), B (other fully halogenated CFCs, carbon tetrachloride, methyl chloroform), C (Hydrochlorofluorocarbons, HCFCs), E (methyl bromide) and F (hydrofluorocarbons, HFCs).
Aim
- With 197 nations party to the accord, it aims to protect the ozone layer by taking measures to control total global production and consumption of ODS, with the ultimate objective of their elimination on the basis of developments in scientific knowledge and technological information.
Universal Ratification
- On 16th September 2009, the Vienna Convention and the Montreal Protocol became the first treaties in the history of the United Nations to achieve universal ratification.
Amendments to the Montreal Protocol
- The London Amendment (1990): It changed the ODS emission schedule by requiring the complete phase out of CFCs, halons, and carbon tetrachloride by 2000 in developed countries, and by 2010 in developing countries. Methyl chloroform was also added to the list of controlled ODSs, with phase out in developed countries targeted in 2005, and in 2015 for developing countries.
- The Copenhagen Amendment (1992): It significantly accelerated the phase out of ODSs and incorporated HCFCphase out for developed countries, beginning in 2004. Under this agreement, CFCs, halons, carbon tetrachloride, and methyl chloroform were targeted for complete phase out in 1996 in developed countries. In addition, methyl bromide consumption of methyl bromide was capped at 1991 levels.
- The Montreal Amendment (1997): It included the phase out of HCFCs in developing countries, as well as the phase out of methyl bromide in developed and developing countries in 2005 and 2015, respectively.
- The Beijing Amendment (1999):It tightened controls on the production and trade of HCFCs. Bromochloromethane was also added to the list of controlled substances with phase out targeted for 2004.
- The Kigali Amendment (2016):It extended controls to phase down the production and consumption of HFCs because these substances were adopted by industries in moving away from ozone-depleting substances and they are potent greenhouse gases damaging to the earth’s climate.
Impact of Montreal Protocol
- It has helped successfully curbed 98% of ozone-depleting substances over the last three decades.
- It has successfully put a stop to the use and emission of CFC (Chlorofluorocarbons) and other harmful gases that continuously worked on thinning the layer of ozone above Earth.
- A very significant co-benefit is that it has - during period 1989-2013 - reduced cumulative CO2-eq. emissions by 135 billion tonnes.
- HFCs replaced the CFCs, predominantly used in cooling appliances. Though the HFCs did not affect the ozone, they have actively added to global warming.
Kigali Amendment,2016
It divided the signatory parties into three groups as follow:
Significance
|
Ozone Layer
- Ozone is created in the atmosphere when the sun's rays split oxygen molecules into single atoms. These atoms combine with nearby oxygen to form a three-oxygen molecule, called ozone.
- The ozone layer is a region of high ozone concentration in the stratosphere, 20 to 30 kilometres above Earth's surface.
- Ozone concentrations are measured in Dobson Units (DU).
Ozone Depleting Substances (ODS)
- Manmade chemicals containing halogens were determined to be the main cause of ozone loss. These chemicals are collectively known as ozone-depleting substances (ODSs).
- The most important are chlorofluorocarbons (CFCs), which at one time were widely used in air conditioners, refrigerators and aerosol cans. Other chemicals, such as hydrochlorofluorocarbons (HCFCs), halons, sterilants, carbon tetrachloride and methyl bromide also deplete the ozone layer.
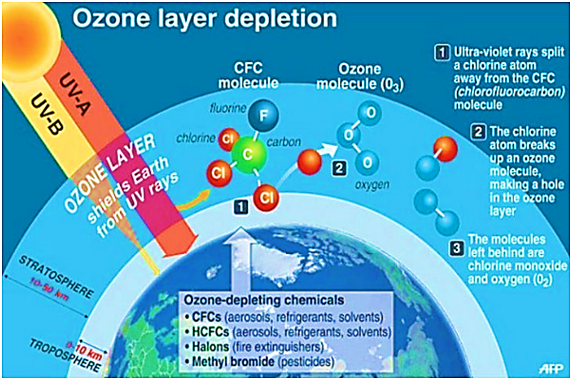
How ODS is causing Harm to Ozone Layer?
- When a CFC molecule reaches the stratosphere, it eventually absorbs UV radiation, causing it to decompose and release its chlorine atoms. One chlorine atom can destroy up to 100,000 ozone molecules.
- Too many of these chlorine and bromine reactions disrupt the delicate chemical balance that maintains the ozone layer, causing ozone to be destroyed faster than it is created.
Importance of Ozone layer
- Ozone in the stratosphere absorbs some of the Sun’s biologically harmful ultraviolet radiation. Because of this beneficial role, stratospheric ozone is considered good ozone.
- In contrast, excess ozone at Earth’s surface that is formed from pollutants is considered harmful because it can be harmful to humans, plants, and animals.
- The amounts of "good" stratospheric and "bad" tropospheric ozone in the atmosphere depend on a balance between processes that create ozone and those that destroy it. An upset in the ozone balance can have serious consequences for life on Earth.



